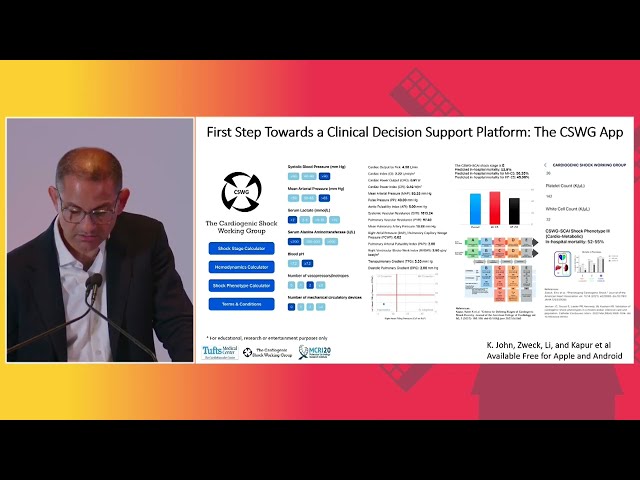
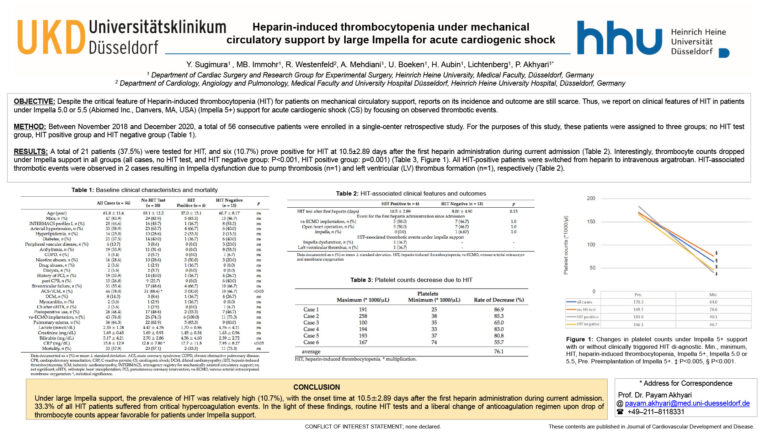
To Have Clinical Value, Future Cardioprotective Interventions Must Provide Additive Protection To That From a P2Y12 Receptor Antagonist
Michael Cohen, James M. Downey
Abstract
Patients with AMI are treated with P2Y12 receptor antagonists to suppress platelet aggregation. Although P2Y12 antagonists are used for their anticoagulant effect, our studies reveal that they are also potent postconditioning-mimetics. The latter properties are influencing pre-clinical and clinical searches for adjunctive cardioprotective interventions in unexpected ways. In situ monkey, rabbit, and rat hearts were subjected to ischemia/reperfusion after which hearts were removed, areas at risk determined, and infarct size measured by triphenyltetrazolium chloride staining. Animals were given cangrelor, an intravenous P2Y12 antagonist, starting 10 min before reperfusion to simulate treatment of patients with AMI prior to angioplasty. Infarction was approximately half that seen in untreated animals. This cardioprotection was abrogated by inhibitors of signaling used by postconditioning including wortmannin, LY294002, PD98059, 5-hydroxydecanoate, 8-sulfophenyltheophylline, MRS1754, and 2-mercaptopropionylglycine suggesting that cangrelor protects by a similar mechanism. None restored platelet reactivity indicating that protection was independent of anti-coagulation. Our hypothesis was further strengthened by failure of ischemic pre- or postconditioning to potentiate cangrelor’s cardioprotection. Similar results were seen with clopidogrel and ticagrelor, other P2Y12 antagonists, indicating a class effect. We propose that much of the ability of P2Y12 inhibitors to improve outcomes in AMI can be attributed to direct infarct size reduction from conditioning rather than prevention of stent thrombosis. Because patients treated with P2Y12 antagonists prior to revascularization are already postconditioned, adding another conditioning intervention will be futile. This would explain why recent large trials of ischemic postconditioning or the postconditioning-mimetic cyclosporine in AMI patients were unsuccessful in spite of strong preclinical results. Additional protection in today’s patients will require an intervention not dependent on the conditioning mechanism. Any such intervention must first demonstrate additive protection in animals that have additionally been treated with a P2Y12 antagonist before clinical trials are considered.